Introduction
Consumers currently expect foods not only to provide satiety and nutrients but also prevent disease and improve health by promoting physical and mental well-being. Accordingly, functional foods are widely used to achieve these effects. The term functional food was first used in Japan and is used to describe foods that contain beneficial ingredients and improve consumer health. Representative functional ingredients include probiotics, prebiotics, vitamins, and minerals. By using these ingredients, functional foods, such as fermented milk, yogurt, and sports drinks, have been developed (Stanton et al., 2001; Siró et al., 2008).
The definition of probiotics was redefined in 2001 by The Food and Agriculture Organization of the United Nations/World Health Organization as “live microorganisms, which when administered in adequate amounts, confer a health benefit to the host. Probiotics must satisfy certain characteristics, including 1) resistance to gastric acidity, 2) bile salt hydrolase activity and resistance to bile salts, 3) adherence to mucus and/or human epithelial cells and cell lines, and 4) antimicrobial and antagonistic activities against potentially pathogenic bacteria (Shokryazdan et al., 2017).
According to the Meeting of the International Scientific Association of Probiotics and Prebiotics (ISAPP) in December 2016, prebiotics are defined as “a substrate that is selectively utilized by host microorganisms conferring a health benefit.” Based on this definition, prebiotics must meet the following criteria: resistance to gastric acidity, hydrolysis by mammalian enzymes, absorption in the upper gastrointestinal tract, ingredients fermented by intestinal microflora, and selective stimulation of intestinal bacteria with positive changes associated with health and well-being (Gibson et al., 2017).
Synbiotics are defined as “a mixture comprising live microorganisms and substrate(s) selectively utilized by host microorganisms that confer a health benefit to the host” (Swanson et al., 2020). The word is derived from the word synergism; hence, it reflects that prebiotics exert a protective effect on probiotics and promote their growth to preserve the population of good bacteria to overcome extreme conditions in the gastrointestinal tract (Markowiak & Śliżewska, 2017). Based on this synergistic principle, the present study aimed to verify whether the combination of Lactiplantibacillus pentosus SC65 and oligosaccharides exhibits these expected benefits. Specifically, we investigated the synergistic effects of this symnbiotic formulation on bacteria viability during storage and its functional potential, such as immunostimulatory activity, compared to singular treatments.
Yogurt is a milk product fermented with Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus, which is usually used as a starter culture (Nagaoka, 2019). During milk fermentation, lactic acid bacteria (LAB) convert lactose to lactic acid by lowering the pH. Consequently, the pH of commercial yogurt usually ranges from 3.7 to 4.3 (Lourens-Hattingh & Viljoen, 2001). Decreasing the pH of milk causes acid coagulation (gelation) of milk proteins, which is mediated by disulfide cross-linking between denatured whey proteins and casein micelles (Corrieu & Béal, 2016). Yogurt is a commonly used dairy product that carries viable bacteria considered as probiotics (Nagaoka, 2019).
Lactiplantibacillus pentosus is widely recognized as a probiotics strain. Notably, L. pentosus strains derived from Kkimchi exhibit acid and bile tolerance, characteristics essential for survival in the intestinal environment (Chu & Kook, 2019; Shin et al., 2021). Specifically, L. pentosus SC65 has been previously reported to enhance immunostimulatory effects through the fermentation of germinated black beans (Lee et al., 2018) and to improve lung health (Lee & Park, 2021).
Therefore, this study aimed to produce synbiotic yogurt using L. pentosus SC65 and fructooligosaccharides (FOS). The bacterium was selected as a potential probiotics in a previous study, and FOS have been widely used as a prebiotic. Overall, the study functionally evaluated the synbiotic yogurt and compared it to yogurt with different combinations during 21 days of cold storage.
Materials and Methods
L. pentosus SC65, isolated from pickled burdock, and Lacticaseibacillus rhamnosus GG (LGG) were stored at −80°C. The bacteria were cultured in de Man, Rogosa and Sharpe (MRS) medium (BD Biosciences, Franklin Lakes, NJ, USA) at 37°C overnight and stored at 4°C for further experiments.
To produce yogurt, skim milk powder (AnchorTM, Auckland, New Zealand) and a commercial starter culture (LYOFAST YCE 438A, SACCO, Cadorago, Italy) consisting of S. thermophilus and Lactobacillus delbrueckii subsp. bulgaricus were used. The L. pentosus SC65 and LGG were used as reference probiotic strains, and FOS from chicory (Fibrelite, Sensus B.V., Roosendaal, Netherlands) was used as a prebiotic.
Prior to yogurt production, sterile 10 mL aliquots of MRS broth were inoculated with 1% (w/v) of each freeze-dried strain. S. thermophilus was incubated at 37°C, and Lactobacillus delbrueckii subsp. bulgaricus was incubated at 42°C. Subsequently, the activated strains were sub-cultured at 1% (v/v) into 10 mL of sterile reconstituted skimmed milk (containing 12% skim milk, 2% glucose, and 1% yeast extract) to serve as the starter inoculum. The milk base (1 L) containing 12% (w/v) skim milk powder was heated at 85°C for 30 min followed by cooling at 42°C in a water bath. Thereafter, the starter culture was inoculated at 0.01% (v/v). Additionally, L. pentosus SC65 and LGG were inoculated at a concentration of 108 colony-forming unit (CFU)/mL. The FOS solution (2% w/v) was sterilized using a 0.22 μm pore size syringe filter (polyvinylidene fluoride, PVDF) before being supplemented. The following six yogurt batches were prepared: S, yogurt supplemented with the starter; LS, starter+LGG; PS, starter+L. pentosus SC65; FS, starter+FOS; FL, starter+FOS+LGG; and FP, starter+FOS+L. pentosus SC65. After inoculation, the mixture was incubated at 42°C until the pH dropped to 4.5±0.1. The yogurt mix was cooled immediately and stored at 4°C for further experiments. All experiments were conducted on storage days 0 (after fermentation), 1, 7, 14, and 21.
Each yogurt sample (100 μL) was suspended in 900 μL of 0.88% NaCl for serial dilution. The diluted samples (100 μL) were then spread onto MRS medium and anaerobically incubated at 37°C for 72 h using the GasPak™ EZ Anaerobe Container System (BD Biosciences).
The pH was measured using a pH meter (FiveEasy Plus; Mettler-Toledo Inc., Columbus, OH, USA). Yogurt (3 g) was diluted with 27 mL of water to measure the titratable acidity. With the addition of 0.5% phenolphthalein solution as a pH indicator, the pH of yogurt was titrated until 8.2±0.1 using 0.1 N NaOH solution. The amount of NaOH (0.1 N) used for titration was expressed as the titratable acidity (%) using the following equation:
where 10 is the dilution factor; VNaOH is the volume of 0.1 N NaOH (mL) consumed, 0.1 is the normality of NaOH solution, and 0.009 is the conversion factor of lactic acid.
Sulfuric acid (0.1 N, 10 mL) was used to dilute 1.6 mL of the yogurt sample to obtain separated lactic acid from the yogurt. The diluted sample was centrifuged at 7,000×g for 5 min at 4°C, and the obtained supernatant was filtered using a 0.22-μm SmartPor®-ll PVDF Syringe filter (Woongki Science Co., Ltd., Seoul, Korea). Lactic acid was detected using high-performance liquid chromatography with a Dionex UltiMate 3000 system, consisting of Ultimate 3000 RS pump, Ultimate 3000 RS autosampler, Ultimate 3000 RS Column Compartment, and Ultimate 3000 Variable Wavelength Detector (Thermo Fisher Scientific, Waltham, MA, USA). Lactic acid was separated using an Aminex HPX-87H (300×7.8 mm) ion-exchange column (Bio-Rad, Hercules, CA, USA). Each sample (10 μL) was injected, and the mobile phase was 0.0075 N H2SO4 with a flow rate of 0.6 mL/min for a runtime of 30 min. The analysis was performed at 65°C, and ultraviolet detection was conducted at 210 nm. A standard curve in the range of 6.25–100 mM of L-lactic acid was used for quantification.
Each sample (10 g) was centrifuged at 600×g for 6 min at 4°C, which consequently resulted in two divided layers. The upper layer was used to assess syneresis, and the water-holding capacity (WHC) was calculated using the weight of the precipitate.
Radical scavenging activity was assessed using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. DPPH solution (0.1 mM) dissolved in methanol was prepared, and 100 μL of yogurt sample was mixed with 900 μL of DPPH solution at a ratio of 1:9 (v/v). The reaction mixture was then incubated at 20°C in the dark for 30 min. After incubation, the precipitate was removed by centrifugation. Clear samples were placed into each well of a 96-well plate, and the absorbance was measured at 517 nm using an Epoch Microplate Spectrophotometer (Bio-Tek Instruments, Winooski, VT, USA). The percentage of DPPH radical scavenging activity was calculated using the following formula:
where the absorbance of the yogurt and blank (negative control, distilled water) were expressed as Asample and Acontrol, respectively.
RAW 264.7 macrophage cells (KCLB 40071) were obtained from Korean Cell Line Bank (Seoul, Korea). Cells were cultured at 37°C with 5% CO2 in high glucose Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco, Grand Island, NY, USA) supplemented with 1% (v/v) penicillin-streptomycin (Gibco, USA) and 10% (v/v) fetal bovine serum (Gibco, USA).
RAW 264.7 macrophage cells were cultured in a 96-well plate at a concentration of 5×104 cells/well. After 20-h incubation, cells were treated with 100 μL of yogurt samples diluted to concentration of 5×106 and 1×107 CFU/mL, or DMEM as a negative control and incubated for 24 h. The plate was washed three times with Dulbecco’s Phosphate Buffered Saline (Welgene, Daegu, Korea) and re-filled with 200 μL of DMEM, followed by a reaction with 20 μL of EZ-CYTOX (DoGenBio, Seoul, Korea) for 25–30 min. The absorbance was measured at 480 nm using an Epoch Microplate Spectrophotometer (Bio-Tek Instruments).
In a 24-well plate, RAW 264.7 cells were seeded at a concentration of 5×104 cells/well in 500 μL of DMEM and incubated at 37°C for 20 h with 5% CO2. The cells were then treated with lipopolysaccharide (LPS, 1 μg/mL) as a positive control or desired concentrations of yogurt samples at a total volume of 500 μL, and DMEM was used as a negative control. After 24 h of incubation, the supernatant (100 μL) was collected in a 96-well plate and mixed with 100 μL of Griess reagent consisting of Griess A (0.1% N-(1-naphtyl)ethylenediamine) and Griess B (5% phosphoric acid containing 1% sulfanilamide) (1:1, v/v). The reaction was performed in a dark room for 15 min, and the absorbance of the mixture was measured at 540 nm using an Epoch Microplate Spectrophotometer (Bio-Tek Instruments). To quantify nitric oxide (NO) content, a standard curve was obtained via dilution of sodium nitrite (Sigma-Aldrich, St. Louis, MO, USA) in the range of 0–250 μM.
All experiments were conducted in triplicate, and results are expressed as mean±standard deviations. Statistical significance of the experimental data was assessed via one-way analysis of variance using SPSS version 25 (IBM Co., Armonk, NY, USA). Tukey’s multiple range analysis was used to determine significant differences between samples or treatments, with significance set at p<0.05.
Results and Discussion
During fermentation, the number of viable bacterial cells was measured, and the average bacterial count during the overall storage was 8.92 log CFU/mL. The number of viable bacterial cells in the yogurts without FOS (S, LS, and PS) decreased markedly from 8.94, 9.15, and 9.05 log CFU/mL to 8.67, 8.82, and 8.99 log CFU/mL, respectively. In yogurts with FOS (FS, FL, and FP), the bacterial count decreased slightly or showed no statistical difference (Fig. 1A). Between 7 and 14 days of storage, when the number of viable bacterial cells rapidly decreased, the yogurts containing FOS showed a relatively smaller decrease than that of the yogurt without FOS. Damin et al. (2008) and Sarvari et al. (2014) reported that during refrigerated storage, L. delbrueckii subsp. bulgaricus, a component of the starter, grows and produces acid, hydrogen peroxide, and possibly bacteriocins that suppress microorganisms. In addition, the acidic conditions in yogurt damage the microbial population (Simões Bandiera et al., 2013). Furthermore, the results of this study, in which FOS alleviated the decrease in bacterial cell viability, corresponded with those of earlier studies. Madhu et al. (2012) reported that synbiotic yogurt containing 1% FOS had more than 9 log CFU/mL after storage for 28 days, which was a higher viable cell bacterial count than that containing only starter and probiotics. Therefore, the number of viable bacterial cells could be improved by supplementing the medium with nutrients and improving the unfavorable environment, thereby enhancing the viability of probiotics. Similar results have been reported by Desai et al. (2004) and Gustaw et al. (2011).
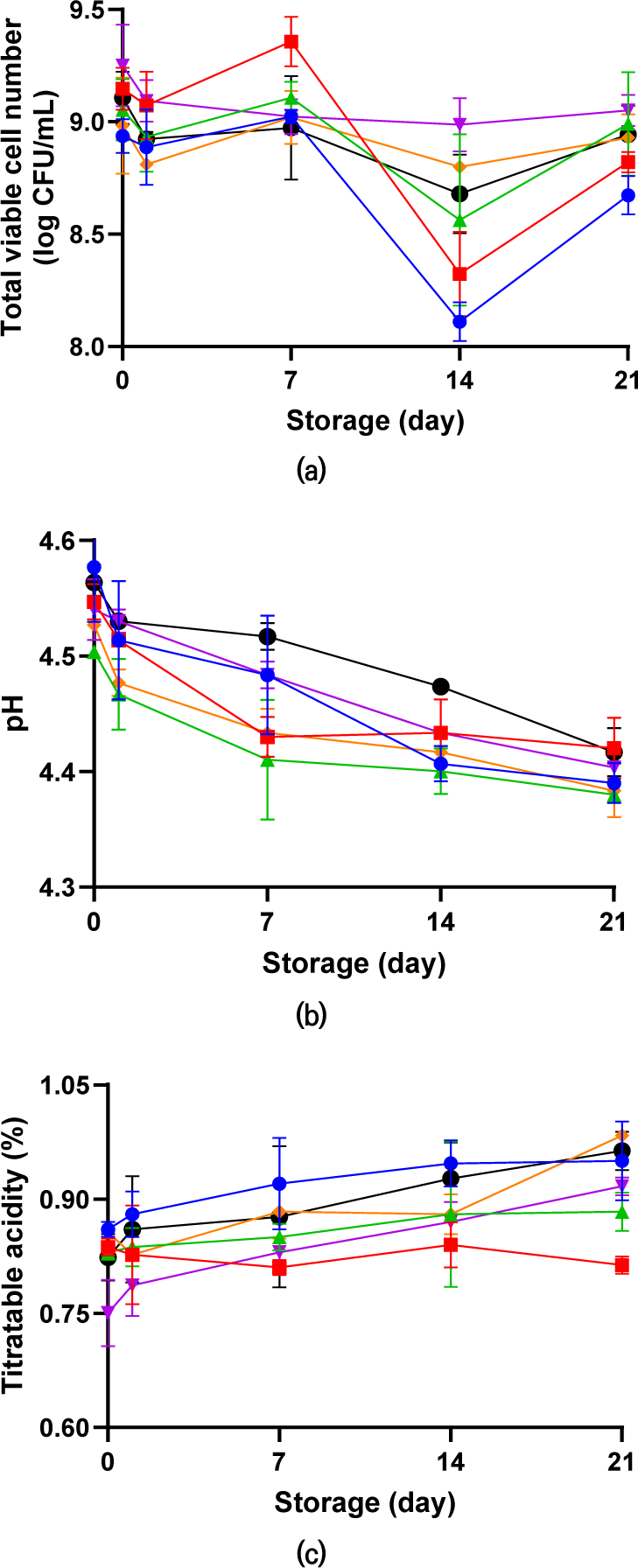
Post-acidification or post-fermentation acidification during storage is attributable to the residual activity of microorganisms. This is an undesired process in yogurt, which increases the size of casein micelles to form new hydrophobic and electrostatic interactions between proteins, thereby causing partial reorganization of the protein network (Guénard-Lampron et al., 2020; Deshwal et al., 2021).
During refrigerated yogurt storage, pH and titratable acidity were measured (Fig. 1B and C). The pH value of all yogurts ranged from 4.50 to 4.58 at the end of fermentation (0 day). Over time, the pH gradually decreased and ranged from 4.38 to 4.42 on the last day. Given that titratable acidity is the opposite property of pH, these values ranged from 0.75% to 0.86 % at the initial stage and from 0.81% to 0.99% on the last day. When comparing yogurts with FOS supplementation, FOS-supplemented yogurts (FS, FL, and FP) showed no statistically significant differences in pH from those of non-supplemented (S, LS, and PS) yogurts at the end of storage. Similar results of oligosaccharides not affecting the acidification of yogurt were described in Madhu et al. (2012), Kariyawasam et al. (2021), and Oliveira et al. (2009), where oligosaccharides led to lower post-acidification.
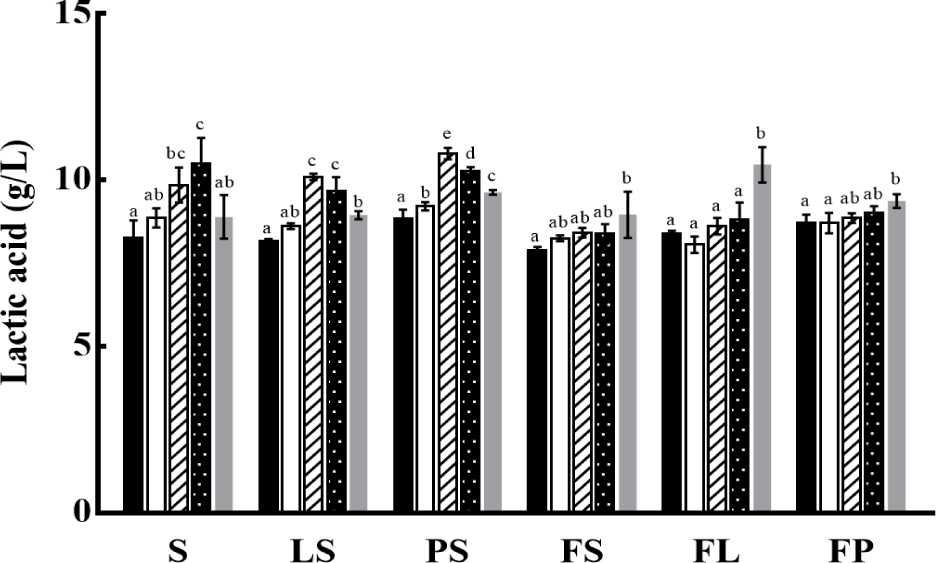
Lactic acid imparts an acidic flavor to yogurt and is derived from carbon sources, such as lactose, glucose, and fructose, in milk. The production of lactic acid continues after fermentation and during storage, which leads to excessive acidification of yogurt and affects the organoleptic characteristics (Chen et al., 2017). The detection and quantification of lactic acid are shown in Fig. 2. Similar to the decrease in pH and increase in titratable acidity, the production of lactic acid increased as the storage period elapsed. Low lactic acid production was also observed in yogurts supplemented with FOS. These results indicated that the addition of FOS was advantageous in preventing post-acidification because it maintained pH stability during storage.
When the capacity of yogurt protein networks to hold water is weakened, syneresis occurs. Syneresis is the release of liquids, such as serum or whey, on the surface of a gel and is regarded as one of the undesirable qualities of yogurt. The main problem with syneresis is the formation of an unstable protein network, which weakens the connections between whey proteins. This requires consideration because undesired sensory characteristics, such as reduced texture or off-flavors resulting from syneresis, may cause consumer dissatisfaction (Bahrami et al., 2013; Arab et al., 2023).
The syneresis and WHC results of the six yogurt samples are shown in Table 1. In S, LS, and PS, the syneresis values were 25.13–36.89% on the initial day but increased during storage to 39.05–46.57%. In contrast, the syneresis values of FOS-supplemented samples (FS, FL, and FP) decreased from 39.67–50.22% to 42.40–44.61%. During the entire storage period, better results were initially obtained for the yogurt without FOS; however, similar syneresis values were eventually observed in yogurt with FOS. In the case of WHC, contrasting results were observed.
Capital letters indicate statistically significant differences between samples on the same day. Lowercase letters indicate statistically significant differences between storage days in the same sample. Data are expressed as mean±standard deviation. S, starter; LS, starter+LGG; PS, starter+Lactiplantibacillus pentosus SC65; FS, starter+FOS; FL, starter+FOS+LGG; FP, starter+FOS+L. pentosus SC65; LGG, Lacticaseibacillus rhamnosus GG; FOS, fructooligosaccharides; WHC, water-holding capacity.
Generally, as the storage time increased, the WHC of the yogurt increased with higher syneresis. However, based on this result, FOS may have positive effects on syneresis during cold storage. Similarly, Pachekrepapol et al. (2021) reported that with the addition of 3% FOS, the syneresis of yogurts could be controlled to a lower degree because the structure of FOS enables it to hold free serum, resulting in an increased WHC value (Pachekrepapol et al., 2021). Similar results have been reported by Aghajani et al. (2014).
The radical scavenging activity of yogurt was determined using the DPPH assay after 21 days of storage (Table 2). Probiotics-containing yogurt samples (LS, PS, FL, and FP) showed higher radical-scavenging activity than that of S and FS. One day after fermentation, the DPPH radical scavenging activities of S and FS were 40.52% and 35.61%, respectively, and increased to 41.96% and 50.99% after 21 days. When comparing the probiotics-containing yogurts, the L. pentosus SC65 addition resulted in greater inhibition than that of LGG.
Capital letters indicate statistically significant differences between different samples on the same day. Lowercase letters indicate statistically significant differences between storage days in the same sample. Data are expressed as mean±standard deviation (SD). S, starter; LS, starter+LGG; PS, starter+Lactiplantibacillus pentosus SC65; FS, starter+FOS; FL, starter+FOS+LGG; FP, starter+FOS+L. pentosus SC65; LGG, Lacticaseibacillus rhamnosus GG; FOS, fructooligosaccharides; DPPH, 2,2-diphenyl-1-picrylhydrazyl.
Shu et al. (2017) investigated the effects of prebiotics, such as inulin, FOS, galactooligosaccharides, and xylooligosaccharides, on DPPH antioxidant activity. The study reported that all prebiotics enhanced the antioxidant activity of goat milk fermented with Lactiplantibacillus plantarum L60. Synbiotic yogurt containing L. plantarum CFR 2194 and Limosilactobacillus fermentum CFR 2192 effectively inhibited DPPH radicals in the presence of FOS. This could be explained by the metabolic end product, which has a potential antioxidant effect resulting from the utilization of FOS by LAB (Shu et al., 2017).
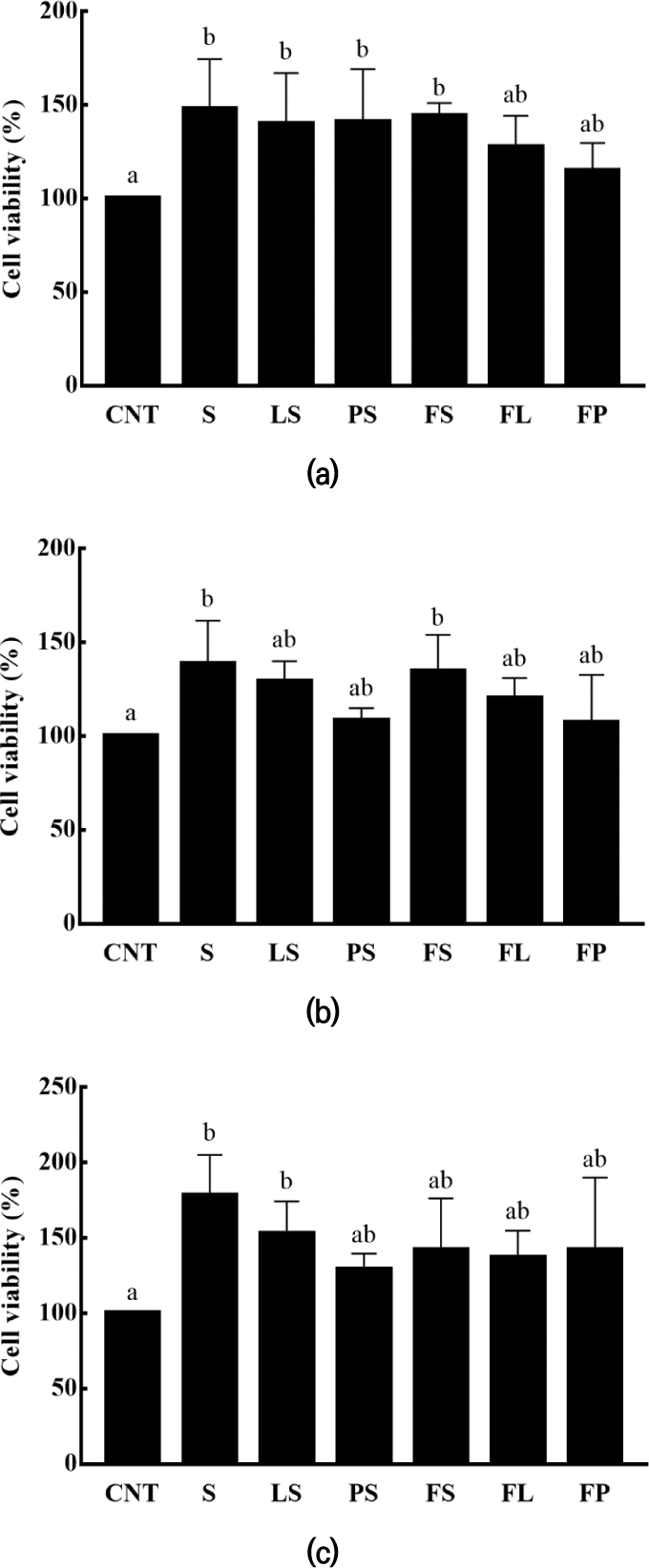
Yogurt improves host defense mechanisms against invading pathogenic agents; however, these mechanisms have not been fully defined. The immunostimulatory effects of yogurt are closely related to its bacterial components (Meydani & Ha, 2000). Considering that probiotic bacteria in yogurt activate macrophages and enhance the immune system (Mishra et al., 2008), the cytotoxicity of yogurt on macrophages was determined before demonstrating its immunostimulatory effects.
RAW 264.7 macrophage cells (5×104 cells/well) were used, and the EZ-cytox assay was employed to measure the cytotoxicity of yogurt. According to 3.1 LAB viability, the number of viable bacterial cells in the yogurts were maintained at approximately 9 log CFU/mL until seven days of storage, and the concentration of LAB was calculated based on this result. Although the treatment of yogurt containing LAB could be a stress agent, all batches achieved a bacterial viability of more than 80% at concentrations of 1.25×106, 2.5×106, and 5×106 CFU/mL. Subsequent experiments conducted within this concentration range confirmed the absence of cytotoxicity (Fig. 3).
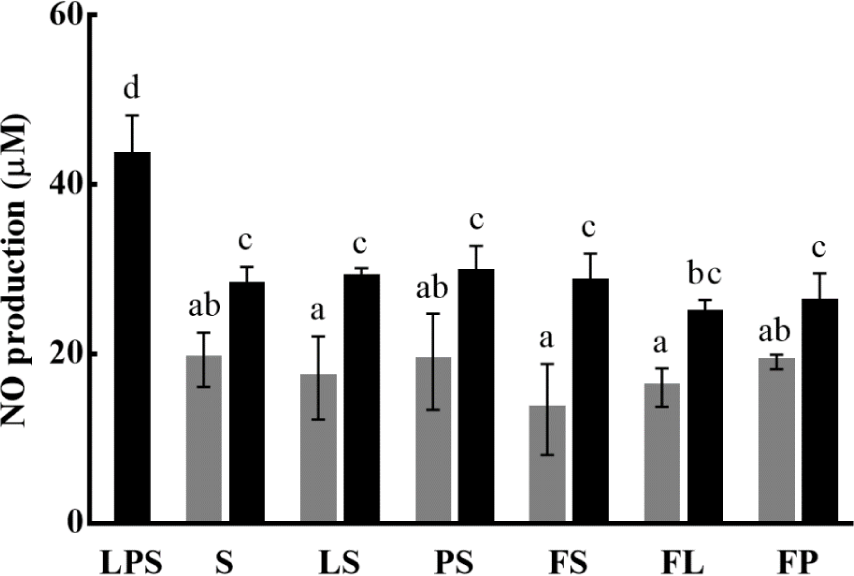
NO is a gaseous free radical molecule that acts as an intra- and intercellular signaling molecule. NO is produced in a reaction catalyzed by nitric oxide synthase (NOS), which results in the oxidative conversion of L-arginine to NO and L-citrulline. Three isoforms of NOS have been reported: endothelial, neuronal NOS, and inducible NOS (iNOS), which is involved in immunometabolism. No reaction occurs in resting cells; however, when pathogenic bacteria or agents, such as LPS, invade cells, NO expression is initiated by iNOS in various inflammatory cells. Because of this reaction, NO protects the host from pathogens by stimulating immune responses (Meyer et al., 2007; Anavi & Tirosh, 2020).
In this study, LPS (1 μg/mL) was used to stimulate the RAW 264.7 macrophage cells as a positive control and resulted in the production of 43.35 μM NO. The potential immunostimulatory effects of the yogurt samples were evaluated by comparison with LPS-stimulated cells (Fig. 4). Based on the cytotoxicity results, 5×106 and 1×107 CFU/mL of each yogurt were used to induce NO production in RAW 264.7 macrophages. When treated with 5×106 CFU/mL of yogurt, 19.31, 17.18, 19.12, 13.45, 16.05, and 19.07 μM NO was produced under treatment with S, LS, PS, FS, FL, and FP, respectively. When FOS was added to the yogurt, no specific effect was observed compared with that in yogurt without FOS. However, in FOS-containing samples, effective production occurred when probiotics, such as LGG and L. pentosus SC65, were added, rather than using only the starter culture containing S. thermophilus and L. bulgaricus. As the concentration increased, NO production also increased. At the treatment concentration of 1×107 CFU/mL of yogurt, the highest production of 29.56 μM NO was observed in sample PS and the lowest concentration of 24.74 μM NO was observed in FL. At this concentration, the amount of NO levels were similar regardless of supplementation with probiotics or FOS.
LGG produces a relatively small amount of NO when used as a reference strain for other LAB. For example, NO production was 2.31 μM under treatment with 1×105 CFU/mL LGG (Yang et al., 2019), 1.61 μM under treatment with 1×106 CFU/mL LGG (Son et al., 2018), and 2.47 μM under treatment with 1×107 CFU/mL LGG (Ahn et al., 2012). Similarly, in the current study, NO production was 0.30 μM and 2.84 μM under treatment with 5×106 CFU/mL and 1×107 CFU/mL LGG, respectively. In contrast, 14.32 μM and 22.79 μM NO was produced when treated with 5×106 CFU/mL and 1×107 CFU/mL L. pentosus SC65, respectively.
LGG is a well-known probiotics with high intestinal adhesion and acid/bile resistance. These characteristics contribute to its beneficial effects, including the prevention of gastrointestinal infections by suppressing respiratory infections, allergies, and pathogenic microorganisms in the intestinal tract (Lim et al., 2013). Furthermore, L. pentosus SC65 was reported as a potential probiotics by Kwon & Park (2021). In this study, both probiotics exhibited higher immune activity after yogurt preparation than when used alone. This increase is considered to have no negative effect on the immunomodulatory effects of each probiotics and is expected to induce a positive effect on host health.
Conclusion
This study aimed to produce synbiotic yogurts and evaluate their functional characteristics through comparative analysis. The following six yogurt samples were prepared: S, yogurt supplemented with starter; LS, starter+LGG; PS, starter+L. pentosus SC65; FS, starter+FOS; FL, starter+FOS+LGG; and FP, starter+FOS+L. pentosus SC65. FOS-supplemented yogurt (FS, FL, and FP) showed a protective effect against syneresis during refrigerated storage. The structure of FOS increases its WHC, which results in the retention of yogurt serum. Thus, the addition of FOS enhanced yogurt preservation. Correspondingly, in terms of functional characteristics, L. pentosus SC65-supplemented yogurt (PS and FP) showed higher DPPH radical scavenging activity than that of the non-LAB-supplemented samples (S and FS). Moreover, all yogurt samples showed concentration-dependent NO production, and NO production was higher than that with probiotics alone. Consequently, L. pentosus SC65 may be a potential candidate ingredient for manufacturing synbiotic yogurt with improved preservation and health benefits.
